Get Educated:
Liver Disease Types & Conditions
Schedule Your Free Liver Scan
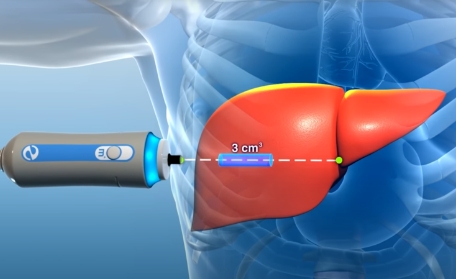
New patients may receive their first FibroScan, free of charge, at any of our Arizona Liver Health locations. A FibroScan is a simple, noninvasive device used for the detection of fat and scarring in the liver.
Request Your Free FibroScan